High-quality leukopaks delivered reliably and ready for your research
We provide fresh and frozen human leukopaks sourced from a diverse pool of healthy, HLA-typed donors. All donors undergo comprehensive screening and testing, including for past CMV infection.
Leukopaks tailored for quality, yield and flexibility
High MNC yield
Stringent quality control
Fully customisable
Unlock new research possibilities
Immunology studies
Investigate immune responses, cell signalling pathways and cytokine production

Cell therapy development
Source material for CAR-T cell production, dendritic cell research and other adoptive cell therapies

Vaccine research
Evaluate vaccine efficacy by studying antigen-specific T cell responses

Flexible formats available to meet your needs
Frozen leukopaks
Cryopreserved using CryoStor® CS10 and a controlled rate freezing system, these provide flexibility for future research use without compromising cell integrity.
Fresh leukopaks
Ideal for immediate use, these are collected and processed on-demand, with next-day delivery available across Europe and same-day options in select UK locations to ensure maximum cell viability.
What our customers say
How to order
Browse our inventory
Access pre-collected, frozen biospecimens from our online catalogue
1. Browse our frozen stock inventory
2. Add samples to your basket
3. Submit your request & reserve the samples
4. Your local distributor will be in touch to confirm
Request a biospecimen collection
Design a custom collection based on your specific research needs
1. Specify your sample, donor & delivery criteria
2. Add to your quote request
3. Submit the request from your basket
4. Your local distributor will be in touch to confirm
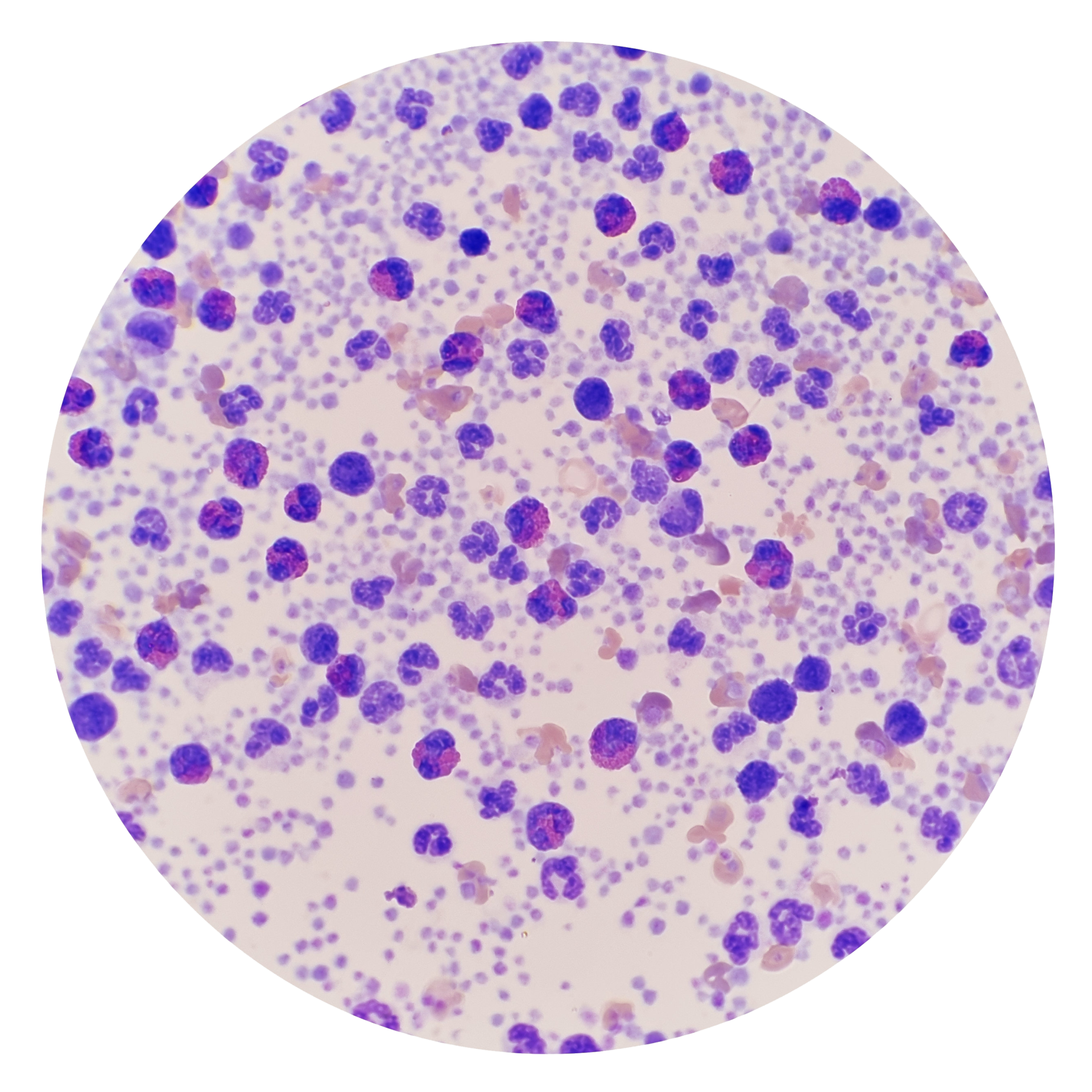
What are leukopaks?
Leukopaks are enriched collections of mononuclear cells (MNCs), obtained through leukapheresis, and contain high numbers of lymphocytes (T cells, B cells, NK cells) and monocytes. These high-yield cell collections are ideal for cell therapy development, immuno-oncology research, and vaccine development.
Right donor.
Right sample.
Right time.
If you can’t see exactly what you need, our team is ready to collaborate on custom collections designed to meet your specific requirements.
Repeat donors for longitudinal studies
HLA-matched and tailored sample collections
Flexible volumes to support large-scale studies or specific applications
Ordering & delivery
We prioritise fast and reliable delivery to maintain sample viability
Same-day delivery options available in select UK locations
Standard next-day delivery across Europe
We're here to help
Whether you need more information about our donor collections or want to
discuss a custom request, our team is ready to assist.


